
 i_need_contribute
i_need_contribute

|
Country, |
Total |
New |
Total |
|
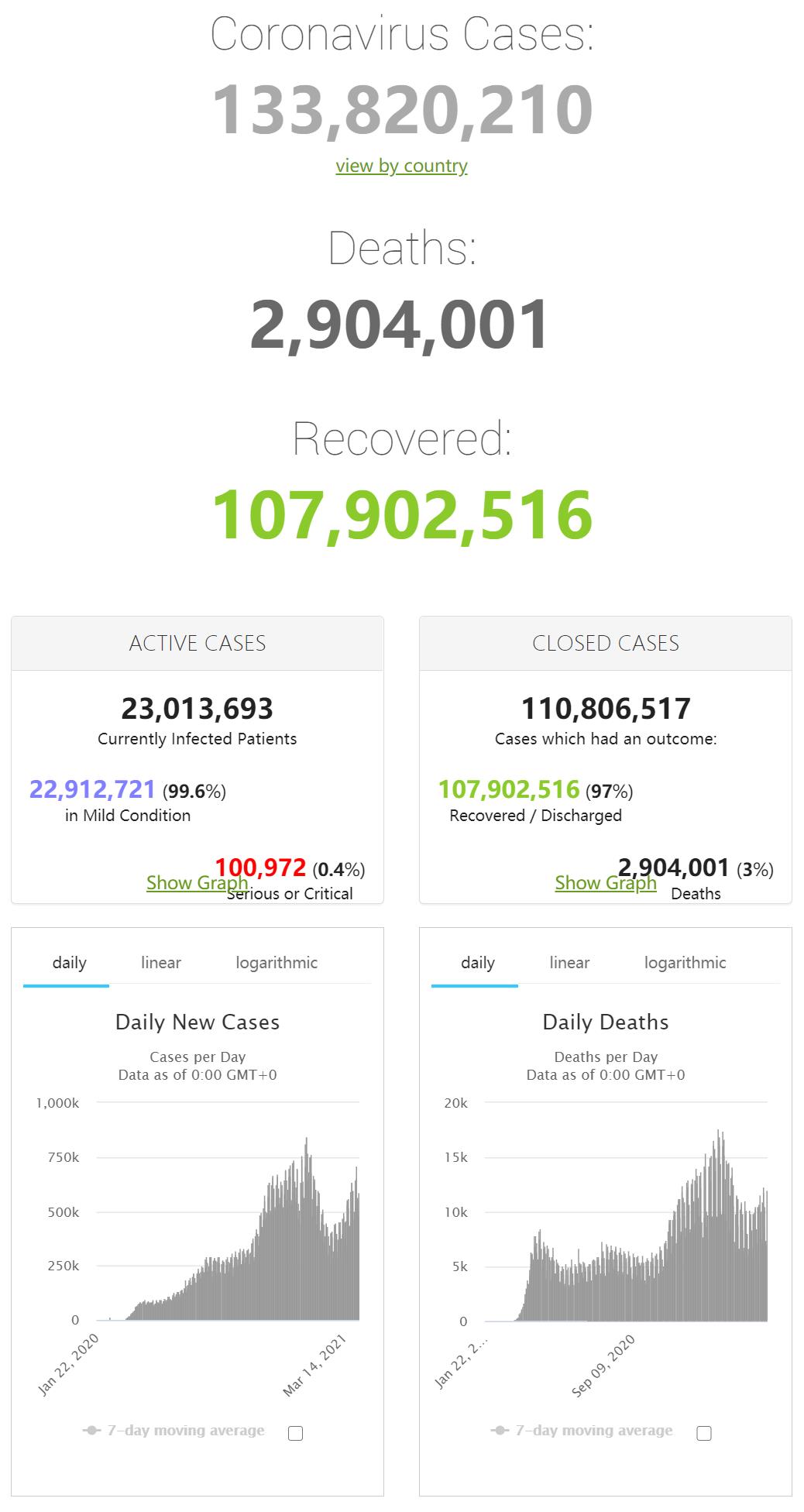
World |
133,675,772 |
+652,169 |
2,900,409 |
|
31,637,243 |
+75,183 |
572,849 |
|
|
13,197,031 |
+90,973 |
341,097 |
|
|
12,926,061 |
+126,315 |
166,892 |
|
|
4,902,985 |
+959 |
97,722 |
|
|
4,606,162 |
+8,294 |
101,480 |
|
|
4,367,291 |
+2,763 |
126,927 |
|
|
3,700,393 |
+13,708 |
112,374 |
|
|
3,633,925 |
+54,740 |
32,943 |
|
|
3,326,736 |
+8,788 |
76,037 |
|
|
2,927,572 |
+14,936 |
78,174 |
|
|
2,479,617 |
+11,381 |
64,767 |
|
|
2,471,617 |
+14,910 |
55,703 |
|
|
2,450,068 |
+22,039 |
56,832 |
|
|
2,256,380 |
+4,675 |
205,002 |
|
|
1,984,348 |
+20,954 |
63,699 |
|
|
1,784,579 |
+15,415 |
35,498 |
|
|
1,607,898 |
+9,305 |
53,725 |
|
|
1,560,833 |
+5,566 |
27,410 |
|
|
1,553,609 |
+756 |
53,111 |
|
|
1,547,376 |
+4,860 |
42,064 |
|
|
1,319,353 |
+6,346 |
16,672 |
|
|
1,043,022 |
+5,242 |
23,796 |
|
|
1,028,041 |
+7,148 |
23,173 |
|
|
988,624 |
+5,407 |
24,561 |
|
|
904,673 |
+1,709 |
23,247 |
|
|
895,622 |
+8,331 |
14,572 |
|
|
835,216 |
+296 |
6,266 |
|
|
825,031 |
+663 |
16,890 |
|
|
819,164 |
+6,414 |
14,059 |
|
|
700,188 |
+4,004 |
15,026 |
|
|
693,676 |
+1,933 |
22,409 |
|
|
659,278 |
+7,626 |
9,447 |
|
|
650,681 |
+5,232 |
7,469 |
|
|
629,388 |
+3,615 |
5,579 |
|
|
566,008 |
+3,101 |
9,546 |
|
|
499,688 |
+663 |
8,867 |
|
|
489,576 |
+2,031 |
9,279 |
|
|
485,918 |
+3,120 |
6,512 |
|
|
476,019 |
+1,883 |
1,520 |
|
|
394,952 |
+783 |
6,719 |
|
|
366,894 |
+1,161 |
10,243 |
|
|
364,419 |
+3,556 |
14,034 |
|
|
357,277 |
+364 |
6,148 |
|
|
354,468 |
+1,139 |
1,304 |
|
|
339,604 |
+1,902 |
17,057 |
|
|
330,536 |
+1,278 |
2,314 |
|
|
285,645 |
+687 |
3,840 |
|
|
285,015 |
+3,445 |
8,607 |
|
|
282,548 |
+1,649 |
6,149 |
|
|
278,768 |
+298 |
3,038 |
|
|
276,890 |
+1,498 |
12,366 |
|
|
276,464 |
+2,595 |
3,780 |
|
|
264,994 |
+1,951 |
9,087 |
|
|
259,133 |
+2,672 |
2,753 |
|
|
258,917 |
+2,080 |
3,156 |
|
|
254,937 |
+334 |
3,369 |
|
|
241,469 |
+1,517 |
1,379 |
|
|
239,325 |
+418 |
4,732 |
|
|
237,718 |
+1,155 |
5,268 |
|
|
234,931 |
+614 |
2,435 |
|
|
227,116 |
+2,380 |
4,584 |
|
|
222,060 |
+1,156 |
3,638 |
|
|
221,890 |
+1,189 |
4,092 |
|
|
221,544 |
+2,163 |
3,058 |
|
|
207,293 |
+783 |
12,290 |
|
|
198,898 |
+1,025 |
3,647 |
|
|
198,374 |
+1,354 |
6,928 |
|
|
192,413 |
+1,277 |
4,686 |
|
|
186,201 |
+940 |
320 |
|
|
170,189 |
+1,115 |
1,705 |
|
|
166,685 |
+1,205 |
1,735 |
|
|
165,287 |
+969 |
2,772 |
|
|
163,498 |
+110 |
2,058 |
|
|
151,931 |
+1,120 |
547 |
|
|
142,541 |
+11 |
3,206 |
|
|
141,365 |
+1,523 |
2,276 |
|
|
137,728 |
+1,302 |
4,073 |
|
|
127,192 |
+256 |
2,291 |
|
|
126,987 |
+3,924 |
1,231 |
|
|
118,004 |
+125 |
3,116 |
|
|
111,664 |
+984 |
976 |
|
|
106,898 |
+668 |
1,756 |
|
|
105,176 |
+643 |
1,946 |
|
|
101,082 |
+1,305 |
683 |
|
|
93,309 |
+279 |
1,334 |
|
|
91,009 |
+63 |
752 |
|
|
90,341 |
+12 |
4,636 |
|
|
89,438 |
+161 |
1,512 |
|
|
89,386 |
+315 |
1,224 |
|
|
84,127 |
+192 |
633 |
|
|
82,601 |
+961 |
442 |
|
|
80,416 |
+348 |
862 |
|
|
68,431 |
+139 |
785 |
|
|
49,366 |
+529 |
266 |
|
|
9,834 |
+74 |
92 |
|
|
9,200 |
+16 |
178 |
|
|
2,659 |
+11 |
35 |
Retrieved from: https://www.worldometers.info/coronavirus/
From CNN’s James Frater

People rest after being vaccinated with the AstraZeneca COVID-19 vaccine at the Brabanthal event center in Heverlee, Belgium, on Wednesday, March 17. Francisco Seco/AP/FILE
Belgium has paused use of the Oxford-AstraZeneca Covid-19 vaccine for people aged 18 to 55 years old, national and regional health ministers announced in a statement.
They said their decision follows a review by the European Medicine Agency (EMA) earlier today, saying there was a “possible link” between the vaccine and rare blood clots.
In their statement, Belgium's Ministers said that based on recent scientific advice, they “have decided to temporarily replace AstraZeneca with another vaccine for people aged 18 to 55 years.”
“All vaccines continue to be used for people aged 56 and over,” the statement reads, emphasizing that the EMA has said the benefits of AstraZeneca still outweigh any risks.
“From the age of 56, the demonstrable benefits of vaccination in preventing hospitalization and mortality are undoubtedly much higher than the consequences of the rare side effects,” it adds.
The Belgian Interministerial Conference on Health will re-evaluate the decision “within 4 weeks.
The statement also adds that Belgium, through the EU conference of health ministers, is requesting that EMA make a more detailed analysis of the benefit-risk analysis per age category and “urgently consider the question regarding the 2nd dose.”
The European Medicines Agency (EMA) said Wednesday that a particular combination of unusual blood clots with low blood platelet counts should be listed as a side effect of the vaccine, but stopped short of recommending its use be limited.
The benefits of the shot outweigh the risks and Covid-19 is a "very serious disease," the EMA added.
From CNN's Richard Greene and Schams Elwazer
The World Health Organization said it was “plausible” there was a link between the AstraZeneca vaccine and rare cases of blood clots.
Having reviewed the latest information from the EU and UK regulators among others, the WHO said “a causal relationship between the vaccine and the occurrence of blood clots with low platelets is considered plausible but is not confirmed.”
In a statement Wednesday, the WHO reiterated that:
“whilst concerning, the events under assessment are very rare, with low numbers reported among the almost 200 million individuals” who have received the vaccine worldwide.
Earlier Wednesday, the European and British medicines regulators announced a “possible link” between the AstraZeneca vaccine and rare cases of blood clots, with the UK announcing it would offer under 30s an alternative vaccine.
From CNN's Esha Mitra in Delhi
A health worker prepares a dose of the Covishield Covid-19 vaccine in Mumbai, India on April 1. Punit Paranjpe/AFP/Getty Images
India's Maharashtra state, where the country's highest new daily coronavirus cases are being reported, has asked the central government to increase vaccine supply amid a shortage, the state's minister of health said.
"Many vaccination centers are having to shut down because they haven’t received sufficient vaccines, people are coming to the centers but our healthcare workers have to tell them that they haven’t received the vaccine so they should go home," Maharashtra's Minister of Health Rajesh Tope told reporters Wednesday.
Tope said he has requested the central Indian authorities to supply more doses of the Covaxin and Covishield vaccines. "In both cities and villages, we have created teams… to bring all those above 45 years old to take the vaccine," he added. "We have had a fast mobilization of recipients but haven’t received enough vaccines."
On Tuesday, Maharashtra state recorded 55,469 new cases, nearly half the new cases across the whole country, according to the state health department. Mumbai, the state capital, recorded 10,030 new cases on Tuesday, according to the city administration.
Kishori Pednekar, the mayor of Mumbai, said the shortage comes at a time when there is rising trust in Covid-19 vaccines. "People want to take the vaccine quickly," she told local media. "We are facing shortages of both Covishield and Covaxin, till yesterday we had only 176000 doses of Covaxin left, at this rate it will become difficult to provide even the second dose of the vaccine to those who have already received the first."
Maharashtra has administered the highest number of vaccine doses of any state, with 8,127,248 people receiving the first dose of the vaccine, the Indian Ministry of Health said on Tuesday.
Second wave grips the country: India on Wednesday reported over 115,000 new cases of coronavirus, the highest single-day total since the pandemic began, according to a CNN tally of figures from the Indian Ministry of Health. The country has seen a rapid surge in Covid-19 cases since March.
Several Indian cities and states have reinstated coronavirus restrictions in the second wave. Maharashtra has imposed weekend lockdowns and curfews, the union territory of Delhi has ordered a night curfew, and in the western state of Gujarat, a night curfew has been implemented in 20 cities.
From Journalists Marcia Reverdosa and Shasta Darlington in Sao Paulo
A nurse prepares a dose of China's Sinovac Covid-19 vaccine at a vaccination center in Rio de Janeiro, on March 31. Mauro Pimentel/AFP/Getty Images
Brazil’s Butantan research institute has temporarily suspended production of the Sinovac CoronaVac Covid-19 vaccine due to a delay of the delivery of raw materials from China, three sources directly connected to the matter told CNN affiliate CNN Brasil Wednesday.
A new shipment of raw materials -- the so called IFA (Active Pharmaceutical Input) ingredient -- scheduled to arrive in Brazil from China on April 9, has now been pushed to April 15, Sao Paulo Gov. Joao Doria said Wednesday during a news conference.
When CNN inquired about the reported suspension of Sinovac’s vaccine production at the Butantan Institute in the state of Sao Paulo, the research center denied the claim but confirmed that it will not produce any new vaccines until the raw materials arrive.
"We didn't suspend production. We are in the process of finalizing the packaging of the material we had previously received. As soon as we receive more raw material we can continue with the process and deliver the missing remaining doses,” Butantan’s press office told CNN Wednesday, adding that “despite the delay” it will meet the contractual deadlines with the country’s health ministry.
The Butantan Institute has delivered 40.7 million out of the 46 million Covid-19 doses promised to be delivered by April 30. The Institute says it will deliver an additional 2.5 million doses next week and told CNN it anticipates the production of Covid-19 vaccines to resume by April 15.
The Covid-19 vaccine raw material (IFA) is supplied by Butantan's Chinese partner Sinovac Biotech. The delay in shipping was caused by the increased campaign of China’s vaccines, according to CNN Brasil.
CNN has reached out to Sinovac Biotech and has not yet received a response.
Retrieved from: https://edition.cnn.com/world/live-news/coronavirus-pandemic-vaccine-updates-04-08-21/index.html
By James Gorman

Scientists have yet to establish how the coronavirus got into human circulation in China. A team of health workers tracing a Covid case in January visited a market stall in Wuhan where the infected person had been.Credit...Gilles Sabrie for The New York Times
The joint international and Chinese mission organized by the World Health Organization on the origins of Covid released its report last week suggesting that for almost every topic it covered, more study was needed. What kind of study and who will do it is the question.
The report suggested pursuing multiple lines of inquiry, focused on the probable origin of the coronavirus in bats. It concluded that the most likely route to humans was through an intermediate animal, perhaps at a wildlife farm. Among future efforts could be surveys of blood banks, to look for cases that could have appeared before December 2019, and tracking down potential animal sources of the virus in wildlife farms, the team proposed.
Critics of the report have sought more consideration of the possibility that a laboratory incident in Wuhan could have led to the first human infection. A loosely organized group of scientists and others who have been meeting online to discuss the possibility of a lab leak released an open letter this week, detailing several ways to conduct a thorough investigation. The letter called for further action, arguing that “critical records and biological samples that could provide essential insights into pandemic origins remain inaccessible.”
Much of the letter echoes an earlier release from the same group detailing what it saw as the failures of the W.H.O. mission. This second letter is more specific in the kind of future investigations it proposes.
The group is seeking a new inquiry that would include biosecurity and biosafety experts, and that could involve the W.H.O. or a separate multinational effort to set up a different process to explore the beginnings of the pandemic and its origins in China.
Jamie Metzl, an author, senior fellow of the Atlantic Council, an international policy think tank and signer of the scientists’ letter, said the renewed calls for a more thorough investigation reflected the need for greater monitoring of and restrictions on the viruses that can be studied in labs around the world.
“This is not about ganging up on China,” Mr. Metzl said.
Mr. Metzl’s group was among those disappointed by the report issued last week, which dismissed out of hand the possibility of a leak from the Wuhan Institute of Virology, calling it extremely unlikely.
The head of the W.H.O., Dr. Tedros Adhanom Ghebreyesus, said later that the mission’s consideration of a possible lab leak was not “extensive enough.”
He continued, “Although the team has concluded that a laboratory leak is the least likely hypothesis, this requires further investigation, potentially with additional missions involving specialist experts, which I am ready to deploy.”
By Sheryl Gay Stolberg and Adam Liptak

A guest showed her “Excelsior Pass,” which offers proof of vaccination, on a phone outside The Shed, a performing and visual arts venue in New York.Credit...Angela Weiss/Agence France-Presse — Getty Images
Around the United States, businesses, schools and politicians are considering “vaccine passports” — digital proof of vaccination against Covid-19 — as a path to reviving the economy and getting Americans back to work and play.
New York has rolled out “Excelsior Pass,” billed by the state as “a free, fast and secure way to present digital proof of Covid-19 vaccination” in case sports and entertainment venues require proof of attendees’ status.
Walmart is offering electronic verification apps to patients vaccinated in its stores so they “can easily access their vaccine status as needed,” the company said.
But the idea is raising legal and ethical questions: Can businesses require employees or customers to provide proof of vaccination when Covid vaccines are ostensibly voluntary?
Can schools require that students prove they have been injected with what is still officially an experimental prophylaxis the same way they require long-approved vaccines for measles and polio? And finally, can governments mandate vaccinations — or stand in the way of businesses or educational institutions that demand proof?
Legal experts say the answer to all of these questions is generally yes, though in a society so divided, politicians are girding for a fight. Government entities like school boards and the Army can require vaccinations for entry, service and travel — practices that flow from a 1905 Supreme Court ruling that said states could require residents to be vaccinated against smallpox or pay a fine.
Backers of digital vaccination cards are pressing the Biden administration to get involved, at least by setting standards for privacy and for verifying the accuracy of the records.
The White House is clearly skittish.
“The government is not now nor will we be supporting a system that requires Americans to carry a credential,” Jen Psaki, the White House press secretary, said on Tuesday.
Conservatives and libertarians, though, are resisting such mandates.
Gov. Brad Little of Idaho, a Republican, signed an executive order on Wednesday barring any governmental entity in the state from requiring vaccine passports. Gov. Ron DeSantis of Florida, also a Republican, signed an executive order on Friday barring businesses from requiring patrons or customers to show vaccine documentation, under penalty of losing state contracts.
And Justin Amash, a former Republican congressman who is now a Libertarian, has suggested that the passports constitute a dangerous intrusion on people’s privacy.
“A vaccine passport — a unified, centralized system for providing or denying access to everyday activities like shopping and dining — would be a nightmare for civil liberties and privacy,” Mr. Amash wrote on Twitter last week.
But, in fact, every state already has a database, or an “immunization registry.” And under “data use agreements,” the states are required to share their registries with the C.D.C., though the agency de-identifies the information and not all states have agreed to provide it.
Retrieved from: https://www.nytimes.com/live/2021/04/07/world/covid-vaccine-coronavirus-cases/first-it-was-masks-now-vaccine-passports-are-dividing-americans

A vaccination center in Kathmandu, Nepal, last month.Credit...Niranjan Shrestha/Associated Press
Three weeks after suspending its vaccination campaign, Nepal has started administering shots again thanks to a gift of doses from China.
Nepal, a poor Himalayan nation, had been depending on vaccines manufactured in neighboring India, but last month India began cutting vaccine exports as the country experienced a surge in coronavirus cases. Nepal’s vaccination effort ground to a halt, even as infections began to rise again.
Last week, Nepal’s other giant neighbor, China, stepped in with a donation of 800,000 doses of the vaccine developed by Sinopharm, a state-owned company.
The vaccines will be administered to essential workers, Nepali students preparing to travel to China to study and those living in districts along the Nepal-China border, health officials said. Taranath Pokhrel, a senior official in Nepal’s health department, said that the Chinese government asked Nepal to give priority to the students and to people involved in cross-border trade, presumably to reduce the risk of infected people crossing into China.
Thousands of Nepali students study at Chinese universities under Chinese government scholarships. China, to increase the appeal of its vaccines, has said that foreigners who are inoculated with Chinese-made vaccines may face fewer bureaucratic hurdles entering the country.
Nepal, a nation of 30 million people, has vaccinated more than 1.7 million and slowly begun reopening to visitors, including to a few hundred climbers attempting to scale Mount Everest. The country reported very few infections in January, but new cases have surpassed 300 in recent days, part of a worrying resurgence in new cases across South Asia. India, which shares a porous border with Nepal, recorded more than 115,000 new infections on Wednesday, by far its highest daily total since the pandemic began.
The future of Nepal’s vaccination campaign remains uncertain because the Chinese donation falls short of the two million vaccine doses Nepal was due to receive under an agreement with the Indian manufacturer, the Serum Institute of India. Nepal officials said that they had paid the company 80 percent of the contract price but received only half of the doses. Serum’s chief executive said this week that he hoped to restart exports by June if new infections in India subsided.
“Our entire diplomatic channels are mobilized to get vaccines, but none has assured us of providing vaccines when we tried to procure them,” Dr. Pokhrel said.
In other news from around the world:
· In Japan, officials in Osaka canceled public Olympic torch relay events scheduled for next week and declared a medical emergency as a surge in coronavirus cases strains the hospital system. The prefecture’s 8.8 million residents were asked not to leave their homes except for essential matters. Olympic organizers said the ceremonial relay would be held at a park without spectators — the latest sign of trouble with the Tokyo Olympics scheduled to open in less than four months.
· The Moderna vaccine is now being administered in Britain, with a 24-year-old woman in Wales who is a caregiver for her grandmother becoming the first person in the country to receive it, on Wednesday. The Pfizer and AstraZeneca shots have already been used in the country. Vaccinations in Britain have slumped this month, reaching their lowest level since the inoculation campaign started. In a Twitter post, Prime Minister Boris Johnson urged people to “get your jab as soon as you are contacted.”
· Regulators in South Korea granted final approval to the Johnson & Johnson vaccine, making it the third vaccine authorized for use in the country amid growing concerns about the pace of its inoculation campaign. Officials reported 668 new coronavirus cases on Wednesday, the highest tally in three months, with most of the cases found in Seoul and other major cities.
· Germany’s troubled vaccine rollout may face another hurdle after a shipment of up to 880,000 Moderna vaccines that had been promised for the end of April was canceled, the news site Business Insider reported. Separately on Wednesday, state and federal health ministers were meeting to discuss how to handle cases of people who have received a first dose of the AstraZeneca vaccine after that shot was discouraged for use in people under 60.